Who We Are
At Context Data Analytics we are senior consulting engineers, with a long view of the fields of 3D vision, robotics and machine learning. We have 30+ years’ experience in research and application development harnessing real sensor data.


Our Focus
Context Data Analytics was created to address the need to apply intelligent algorithms to create precise and robust solutions for real-world problems.
Smart Medical Monitoring
Individual-specific precision medical monitoring.
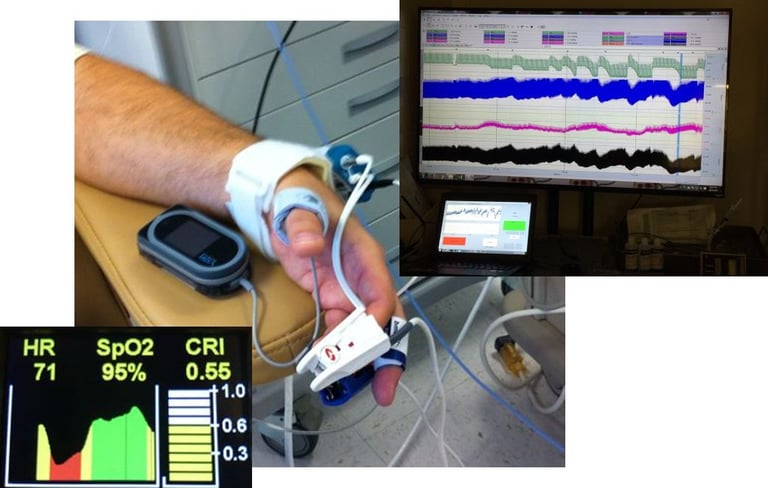
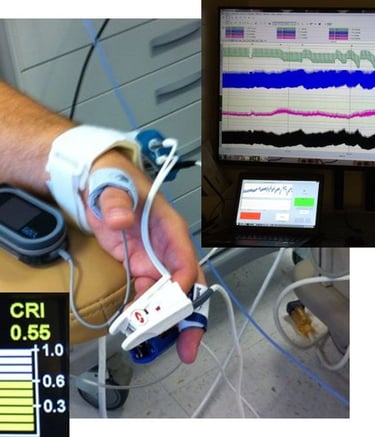
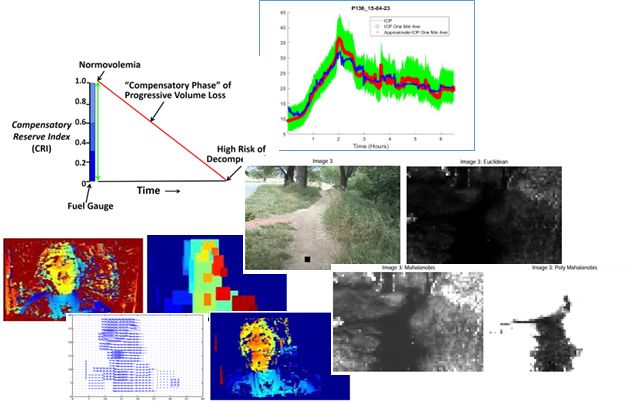
Compensatory Reserve Index (CRI)
Compensatory Reserve Index (CRI) is an FDA cleared algorithm using noninvasive pulsatile continuous vital signals (PPG, ECG, CBP) to detect and monitor hemorrhage and decompensation. The index value takes the range [0-1] trending intravascular volume proximity to hemodynamic decompensation (CRI=0), where CRI=1 is the normal, well hydrated state.
Traditional vital signs only deteriorate when blood loss is already acute
Individuals compensate to different degrees and will collapse after individual-specific blood volume loss.
CRI trends patient specific tolerance to blood loss
Novel tool to detect occult bleeding and monitor resuscitation.
Implications for monitoring other conditions including dehydration and POTS
Developed with DoD funding as a lightweight, point of injury tool to triage and treat patients in the field.
Patented and FDA cleared technology.
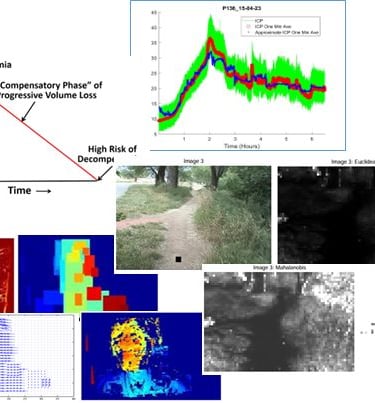
Minimally invasive intracranial pressure monitor developed for porcine model.
Intracranial pressure (ICP) is typically monitored using a bolt (hole bored in skull).
The ability to detect and estimate elevated pressure with less invasive technology could avoid significant risk to patients.
Porcine experiments applied focal brain injury to simulate a mass lesion, while monitoring invasive ICP as well as invasive ABP, ECG and PPG.
Continuous BP-based algorithm estimated ICP with RMSE 2.1 mmHg and cerebral perfusion pressure (CPP) with RMSE 2.7 mmHg.
Noninvasive BP devices or other vital signals could offer noninvasive ICP
Comprehensive human ICP-BP signal data collection and analysis required for product development and FDA submission.